
- J j thomson cathode ray experiment pdf#
- J j thomson cathode ray experiment android#
- J j thomson cathode ray experiment software#
- J j thomson cathode ray experiment Pc#
- J j thomson cathode ray experiment professional#
Ernest Rutherford (left) was awarded Nobel Prize in Chemistry in 1908 for his work in radioactivity. Here the symbol  \(_Z^XA\) is used where Z = atomic number and X = atomic mass of the element A. Figure 7. Thomson measured the charge-by-mass-ratio (e/m) of cathode ray particle using deflection in both electric and magnetic field. Deflection of cathode rays towards positively charged plates Hence cathode ray consists of negatively charged particles. Applying electric field in the path of cathode ray deflects the ray towards positively charged plate. They are independent of the material composition of the cathode.ģ. The schematic of a cathode ray tube is given in figure 2. This ray is called cathode ray and the whole construction is called cathode ray tube. He detected that a stream of particle (ray) was coming out from the negatively charged electrode (cathode) to positively charged electrode (anode). Then he applied a high electrical voltage between two electrodes at either end of the tube. much of the air was pumped out of the tube. Thomson constructed a glass tube which was partially evacuated i.e. Thomson also won the Nobel Prize in 1937 for proving the wavelike properties of electron.
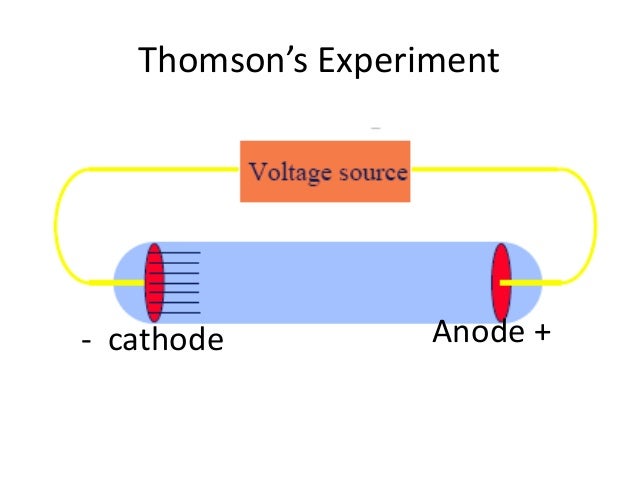
Thomson won Nobel Prize in 1906 for discovering the elementary particle electron. Thomson in 1897 when he was studying the properties of cathode ray.
J j thomson cathode ray experiment professional#
Scholarly yet accessible to those with basic physics knowledge, this book should be of interest to historians of science, professional scientists and engineers, teachers and students of physics, and general readers interested in the development of modern physics.Discovery of Electron Who discovered the electron?Įlectron was discovered by J. The author also focuses on the controversies over N-rays, Becquerel's positive electron, and the famous Ehrenhaft-Millikan dispute over subelectrons.
It explores the events leading to Millikan's unambiguous isolation of the electron and the simultaneous circumstances surrounding the birth of Ernest Rutherford's nuclear atom and the discovery of radioactivity in 1896. It also provides an overview of the history of modern physics up to the advent of the old quantum theory around 1920.Ĭonsolidating scholarly material while incorporating new material discovered by the well-respected author, the book covers the continental and English race for the source of the cathode rays, culminating in Thomson's corpuscle in 1897. The book traces the evolution of the concept of electrical charge, from the earliest glow discharge studies to the final cathode ray and oil drop experiments of J.J. Thomson's Electron presents the compelling story of the discovery of the electron and its role as the first subatomic particle in nature. Flash of the Cathode Rays: A History of J.J. The electron is fundamental to almost all aspects of modern life, controlling the behavior of atoms and how they bind together to form gases, liquids, and solids.
J j thomson cathode ray experiment Pc#
For both formats the functionality available will depend on how you access the ebook (via Bookshelf Online in your browser or via the Bookshelf app on your PC or mobile device).
J j thomson cathode ray experiment pdf#
Where the content of the eBook requires a specific layout, or contains maths or other special characters, the eBook will be available in PDF (PBK) format, which cannot be reflowed. Most VitalSource eBooks are available in a reflowable EPUB format which allows you to resize text to suit you and enables other accessibility features.
